Redox Chemistry - Organic Chemistry
Card 1 of 188
What is the product of the reaction shown?

What is the product of the reaction shown?
Tap to reveal answer
PCC can be used to oxidize primary alcohols into aldehydes, or secondary alcohols into ketones. The starting material shown is a secondary alcohol, so the product will be a ketone (a carbonyl ( ) group where the carbonyl carbon is also attached to two other carbons).
) group where the carbonyl carbon is also attached to two other carbons).
PCC can be used to oxidize primary alcohols into aldehydes, or secondary alcohols into ketones. The starting material shown is a secondary alcohol, so the product will be a ketone (a carbonyl (
← Didn't Know|Knew It →
What is the product when 1-propanol is treated with potassium permanganate?
What is the product when 1-propanol is treated with potassium permanganate?
Tap to reveal answer
Potassium permanganate is a strong oxidizing agent. It can convert secondary alcohols to ketones. It can also convert primary alcohols to carboxylic acids. 1-propanol has a hydroxy group on carbon 1, so it is primary; thus it will be converted to propanoic acid.
Potassium permanganate is a strong oxidizing agent. It can convert secondary alcohols to ketones. It can also convert primary alcohols to carboxylic acids. 1-propanol has a hydroxy group on carbon 1, so it is primary; thus it will be converted to propanoic acid.
← Didn't Know|Knew It →
What is the product of the reaction between magnesium and any alkyl halide, in anhydrous ether?
What is the product of the reaction between magnesium and any alkyl halide, in anhydrous ether?
Tap to reveal answer
The reaction between magnesium and an alkyl halide in anhydrous ether results in a Grignard reagent.
An organolithium would result from the same process, but the magnesium would need to be replaced by two equivalents of lithium. Alcohols are products of reactions between a Grignard reagent and a carbonyl.
The reaction between magnesium and an alkyl halide in anhydrous ether results in a Grignard reagent.
An organolithium would result from the same process, but the magnesium would need to be replaced by two equivalents of lithium. Alcohols are products of reactions between a Grignard reagent and a carbonyl.
← Didn't Know|Knew It →

What type of reaction would ensue if the ketone compound shown was introduced to  (a Grignard reagent in water).
(a Grignard reagent in water).
What type of reaction would ensue if the ketone compound shown was introduced to 
Tap to reveal answer
Grignard reagents are known for their ability to readily attack carbonyls at the point of their carbons. However, Grignard reagents do not work in the presence of protic solvents. Rather than reacting with the desired molecule, the Grignard is so unstable that it will readily accept a proton from a protic solvent. The Grignard then becomes inert and no reaction ensues with the desired molecule.
Grignard reagents are known for their ability to readily attack carbonyls at the point of their carbons. However, Grignard reagents do not work in the presence of protic solvents. Rather than reacting with the desired molecule, the Grignard is so unstable that it will readily accept a proton from a protic solvent. The Grignard then becomes inert and no reaction ensues with the desired molecule.
← Didn't Know|Knew It →

What reactant(s) is/are needed to drive this reaction?
What reactant(s) is/are needed to drive this reaction?
Tap to reveal answer
The carbons on the epoxide compound experience a slightly positive charge. As a result, a Gringard reagent can easily attack the less substituted side of the epoxide to break the ring and to form a six membered carbon chain.  is used to protonate the negatively charged oxygen atom.
is used to protonate the negatively charged oxygen atom.

The carbons on the epoxide compound experience a slightly positive charge. As a result, a Gringard reagent can easily attack the less substituted side of the epoxide to break the ring and to form a six membered carbon chain. 
← Didn't Know|Knew It →

What reagents are needed to satisfy the given reaction?
What reagents are needed to satisfy the given reaction?
Tap to reveal answer

This problem requires that we convert our ketone group into a chlorine. However, this cannot be done directly, and requires multiple steps.
We begin by reducing the ketone with  to form an alcoxide. The alcoxide undergoes workup (the process whereby a negatively charged oxygen gains a proton) via
to form an alcoxide. The alcoxide undergoes workup (the process whereby a negatively charged oxygen gains a proton) via  , depicted above as simply "
, depicted above as simply " ". We now have a secondary alcohol. From here, we can simply use the reagent
". We now have a secondary alcohol. From here, we can simply use the reagent  to convert the alcohol into the desired chlorine.
to convert the alcohol into the desired chlorine.
This problem requires that we convert our ketone group into a chlorine. However, this cannot be done directly, and requires multiple steps.
We begin by reducing the ketone with 



← Didn't Know|Knew It →
Which of the following can be reduced when mixed with  ?
?
Which of the following can be reduced when mixed with 
Tap to reveal answer
 is a very powerful reducing agent that works to reduce almost any carbonyl compound.
is a very powerful reducing agent that works to reduce almost any carbonyl compound.  is an amide and the only carbonyl compound given of the answer choices.
is an amide and the only carbonyl compound given of the answer choices.


← Didn't Know|Knew It →
What is the product of the given reaction?


What is the product of the given reaction?


Tap to reveal answer
First step: esterification
Second step: lithium aluminum hydride reduction
Third step: neutralization to form primary alcohol
Fourth step: SN2 reaction to form final chlorinated product
First step: esterification
Second step: lithium aluminum hydride reduction
Third step: neutralization to form primary alcohol
Fourth step: SN2 reaction to form final chlorinated product
← Didn't Know|Knew It →
What is the result of the following reaction?

What is the result of the following reaction?
Tap to reveal answer
Lithium Aluminum Hydride is a potent reducing agent; it has the ability to turn esters and aldehydes into primary alcohols, and ketones into secondary alcohols. The starting material is an aldehyde, so the correct answer is thus a primary alcohol ONLY.
Lithium Aluminum Hydride is a potent reducing agent; it has the ability to turn esters and aldehydes into primary alcohols, and ketones into secondary alcohols. The starting material is an aldehyde, so the correct answer is thus a primary alcohol ONLY.
← Didn't Know|Knew It →
Which of the following is not a reducing agent?
Which of the following is not a reducing agent?
Tap to reveal answer
 is not a reducing agent; peroxides (compounds with the formula R-O-O-R) are oxidizing agents. A very common peroxide is sodium peroxide
is not a reducing agent; peroxides (compounds with the formula R-O-O-R) are oxidizing agents. A very common peroxide is sodium peroxide  .
.
All of the other listed compounds are reducing agents.


All of the other listed compounds are reducing agents.
← Didn't Know|Knew It →
A water molecule is converted to hydrogen peroxide through a series of reactions. What can you conclude about the oxygen molecule?
A water molecule is converted to hydrogen peroxide through a series of reactions. What can you conclude about the oxygen molecule?
Tap to reveal answer
Water,  , has an oxygen atom with an oxidation number of
, has an oxygen atom with an oxidation number of  whereas hydrogen peroxide,
whereas hydrogen peroxide,  , has an oxygen atom with an oxidation number of
, has an oxygen atom with an oxidation number of  . A less negative oxidation number suggests that the oxygen lost an electron. Recall that oxidation is the loss of electrons whereas reduction is the gain of electrons; therefore, the oxygen atom lost an electron and was oxidized when water was converted to hydrogen peroxide.
. A less negative oxidation number suggests that the oxygen lost an electron. Recall that oxidation is the loss of electrons whereas reduction is the gain of electrons; therefore, the oxygen atom lost an electron and was oxidized when water was converted to hydrogen peroxide.
Water, 



← Didn't Know|Knew It →

Which of the following reagents would satisfy the given reaction?
Which of the following reagents would satisfy the given reaction?
Tap to reveal answer
In order to drive the reactant, we must first convert the alcohol group on the ethanol into a carboxylic acid. We do so by using the oxidizing agent,  , a very strong oxidizing agent that is well known to oxidize primary alcohols into carboxylic acids (among other functions). Once we have our carboxylic acid, we can simply use
, a very strong oxidizing agent that is well known to oxidize primary alcohols into carboxylic acids (among other functions). Once we have our carboxylic acid, we can simply use  to convert our carboxylic acid into an acid halide to attain our desired final product.
to convert our carboxylic acid into an acid halide to attain our desired final product.
In order to drive the reactant, we must first convert the alcohol group on the ethanol into a carboxylic acid. We do so by using the oxidizing agent, 

← Didn't Know|Knew It →
Which reagents reduce alkynes to trans alkenes?
Which reagents reduce alkynes to trans alkenes?
Tap to reveal answer
 produces a trans-alkene from an alkyne whereas
produces a trans-alkene from an alkyne whereas 
 produces a cis-alkene.
produces a cis-alkene.  reduces an alkyne all the way down to an alkane.
reduces an alkyne all the way down to an alkane.  is a strong oxidizing agent.
is a strong oxidizing agent.





← Didn't Know|Knew It →
Which of the following substrates will be oxidized into a ketone when reacting with 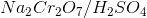 ?
?
Which of the following substrates will be oxidized into a ketone when reacting with 
Tap to reveal answer
 is a strong oxidizing agent.
is a strong oxidizing agent.
Not only can  reduce secondary alcohols into ketones, but it can reduce primary alcohols and aldehydes into carboxylic acids.
reduce secondary alcohols into ketones, but it can reduce primary alcohols and aldehydes into carboxylic acids.

Not only can 
← Didn't Know|Knew It →
3-bromopropene was treated with 
What is the final major product?
3-bromopropene was treated with
What is the final major product?
Tap to reveal answer
Any time we have a Grignard reagent and a primary haloalkane, we will see a substitution reaction, identical to an  reaction. In this case, the Grignard can easily attack the haloalkane as the bromine leaves to create hexene.
reaction. In this case, the Grignard can easily attack the haloalkane as the bromine leaves to create hexene.
Any time we have a Grignard reagent and a primary haloalkane, we will see a substitution reaction, identical to an 
← Didn't Know|Knew It →
Compared to oxygen in water, the oxygen in hydrogen peroxide has valence electron(s).
Compared to oxygen in water, the oxygen in hydrogen peroxide has valence electron(s).
Tap to reveal answer
To solve this question we need to calculate the oxidation number of oxygen in both molecules. The formula for water is  . The oxidation number of hydrogen is +1. Since there are two of them, the hydrogen atoms contribute to a charge of +2. The water molecule is neutral; therefore, the oxygen must have an oxidation number of
. The oxidation number of hydrogen is +1. Since there are two of them, the hydrogen atoms contribute to a charge of +2. The water molecule is neutral; therefore, the oxygen must have an oxidation number of  to balance the charge. The formula for hydrogen peroxide is
to balance the charge. The formula for hydrogen peroxide is  . Using the same logic as water, we can determine that hydrogen contributes +2. We have two oxygen atoms in this case; therefore, each oxygen atom will have an oxidation number of
. Using the same logic as water, we can determine that hydrogen contributes +2. We have two oxygen atoms in this case; therefore, each oxygen atom will have an oxidation number of  to give a charge of
to give a charge of  . This will balance the charges and provide a neutral hydrogen peroxide molecule.
. This will balance the charges and provide a neutral hydrogen peroxide molecule.
Recall that have a negative charge suggests that an atom has extra valence electrons. A charge of  suggests one extra valence electron and a charge of
suggests one extra valence electron and a charge of  suggests two extra valence electrons. An oxygen typically has six valence electrons. The oxygen in water has
suggests two extra valence electrons. An oxygen typically has six valence electrons. The oxygen in water has  oxidation number; therefore, it will have two extra valence electrons (eight total). On the other hand, oxygen in hydrogen peroxide will have one extra valence electron (seven total); therefore, oxygen in hydrogen peroxide has one less valence electron than oxygen in water.
oxidation number; therefore, it will have two extra valence electrons (eight total). On the other hand, oxygen in hydrogen peroxide will have one extra valence electron (seven total); therefore, oxygen in hydrogen peroxide has one less valence electron than oxygen in water.
To solve this question we need to calculate the oxidation number of oxygen in both molecules. The formula for water is 




Recall that have a negative charge suggests that an atom has extra valence electrons. A charge of 


← Didn't Know|Knew It →
Which of the following is true regarding the correct oxidation number of potassium in potassium bromide?
Which of the following is true regarding the correct oxidation number of potassium in potassium bromide?
Tap to reveal answer
Potassium bromide has a formula of  . This molecule is made up of an alkali metal (potassium) and a halogen (bromine). Alkali metals have one valence electron that they readily lose to obtain octet whereas halogens have seven valence electrons and they readily gain an electron to obtain octet. Recall that losing an electron will give you a
. This molecule is made up of an alkali metal (potassium) and a halogen (bromine). Alkali metals have one valence electron that they readily lose to obtain octet whereas halogens have seven valence electrons and they readily gain an electron to obtain octet. Recall that losing an electron will give you a  oxidation number whereas gaining an electron will give you a
oxidation number whereas gaining an electron will give you a  oxidation number. This means that alkali metals always have an oxidation number of
oxidation number. This means that alkali metals always have an oxidation number of  whereas halogens always have an oxidation number of
whereas halogens always have an oxidation number of  ; therefore, potassium has an oxidation number of
; therefore, potassium has an oxidation number of  .
.
Potassium bromide has a formula of 





← Didn't Know|Knew It →
A chemist adds the orange oxidizing agent, Na2Cr2O7, to the following substrates and dissolves the mixture in an aqueous solution of sulfuric acid. Oxidation is indicated by the disappearance of the orange color. Which of the substrate-oxidant solutions will remain orange?
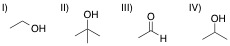
A chemist adds the orange oxidizing agent, Na2Cr2O7, to the following substrates and dissolves the mixture in an aqueous solution of sulfuric acid. Oxidation is indicated by the disappearance of the orange color. Which of the substrate-oxidant solutions will remain orange?
Tap to reveal answer
The following reaction schemes show the oxidation of all substrates, indicating that substrate II is in the highest oxidation state possible, and that an oxidation of this compound will not proceed.
Remember that in sulfuric acid and water, Na2Cr2O7 will be converted to CrO3, the active oxidant species. Furthermore, the oxidation mechanism involving this species includes the key step in which a hydrogen bonded to the carbon in question is eliminated_,_ and simutaneously, a double bond from that carbon to an oxygen is installed. Thus, all substrates that feature at least one hydrogen bonded to the carbon to be oxidized can and will be oxidized in the precense of chromium trioxide.
Lastly, remember that these reactions are taking place in the prescence of water. While substrates such as compound III do not appear to be oxidizable, attack of water at the aldehyde carbon will give a dialcohol tetrathedral intermediate that can be immediately oxidized by chromium trioxide to the corresponding carboxylic acid. A similar mechanism occurs for substrate I, wherein, after the ketone oxidation state is achieved, an attack of water furnishes the same dialcohol intermediate that is oxidized to the carboxylic acid. Remember that the highest oxidation state available for organic compounds containing more than one carbon is the carboxylic acid oxidation state. Chromium trioxide will oxidize all organics to this oxidation state, unless directly-bonded hydrogens are not present in lower oxidation states, such as shown with substrate IV.

The following reaction schemes show the oxidation of all substrates, indicating that substrate II is in the highest oxidation state possible, and that an oxidation of this compound will not proceed.
Remember that in sulfuric acid and water, Na2Cr2O7 will be converted to CrO3, the active oxidant species. Furthermore, the oxidation mechanism involving this species includes the key step in which a hydrogen bonded to the carbon in question is eliminated_,_ and simutaneously, a double bond from that carbon to an oxygen is installed. Thus, all substrates that feature at least one hydrogen bonded to the carbon to be oxidized can and will be oxidized in the precense of chromium trioxide.
Lastly, remember that these reactions are taking place in the prescence of water. While substrates such as compound III do not appear to be oxidizable, attack of water at the aldehyde carbon will give a dialcohol tetrathedral intermediate that can be immediately oxidized by chromium trioxide to the corresponding carboxylic acid. A similar mechanism occurs for substrate I, wherein, after the ketone oxidation state is achieved, an attack of water furnishes the same dialcohol intermediate that is oxidized to the carboxylic acid. Remember that the highest oxidation state available for organic compounds containing more than one carbon is the carboxylic acid oxidation state. Chromium trioxide will oxidize all organics to this oxidation state, unless directly-bonded hydrogens are not present in lower oxidation states, such as shown with substrate IV.

← Didn't Know|Knew It →

What would be the product of the given reaction?

What would be the product of the given reaction?
Tap to reveal answer
The reaction given would give a ketone. This type of reaction is called an oxidation reaction. Oxidation of a secondary alcohol as in the reaction given by  (sodium dichromate) in an aqueous solution of
(sodium dichromate) in an aqueous solution of  (acetic acid) solvent yields a ketone. However, if we performed the same reaction with a primary alcohol, a carboxylic acid would have formed.
(acetic acid) solvent yields a ketone. However, if we performed the same reaction with a primary alcohol, a carboxylic acid would have formed.
The reaction given would give a ketone. This type of reaction is called an oxidation reaction. Oxidation of a secondary alcohol as in the reaction given by 

← Didn't Know|Knew It →
Which of the following reagents can turn primary alcohols into a carboxylic acid?
Which of the following reagents can turn primary alcohols into a carboxylic acid?
Tap to reveal answer
The Jones reagent can convert primary alcohol to acids and secondary alcohols to ketones. The Tollen's test only converts aldehydes to carboxylic acids. PCC can only convert primary and secondary alcohol to aldehydes and ketones, respectively.  and
and  are reducing agents.
are reducing agents.
The Jones reagent can convert primary alcohol to acids and secondary alcohols to ketones. The Tollen's test only converts aldehydes to carboxylic acids. PCC can only convert primary and secondary alcohol to aldehydes and ketones, respectively. 

← Didn't Know|Knew It →