General Chemistry - MCAT Chemical and Physical Foundations of Biological Systems
Card 1 of 3171
Consider the following reaction.

What is the oxidizing reagent in the reaction?
Consider the following reaction.
What is the oxidizing reagent in the reaction?
Tap to reveal answer
The oxidizing reagent is the reactant, not the atom, that is responsible for receiving electrons from another atom in order to oxidize it. The oxygen gas goes from having an oxidation number of 0 to –2. This means that oxygen gas is reduced, but because it oxidized another atom by taking its electrons, we call it the oxidizing reagent.
Reactant oxygen is elemental, and has oxidation number 0.
Product oxygen is in H2O, with net charge of 0.
Total charge = 2(H) + (O) = 0
2(+1) + (O) = 2 + (O) = 0
O = –2
Decrease in oxidation number indicates reduction, and also identifies the oxidizing agent.
The oxidizing reagent is the reactant, not the atom, that is responsible for receiving electrons from another atom in order to oxidize it. The oxygen gas goes from having an oxidation number of 0 to –2. This means that oxygen gas is reduced, but because it oxidized another atom by taking its electrons, we call it the oxidizing reagent.
Reactant oxygen is elemental, and has oxidation number 0.
Product oxygen is in H2O, with net charge of 0.
Total charge = 2(H) + (O) = 0
2(+1) + (O) = 2 + (O) = 0
O = –2
Decrease in oxidation number indicates reduction, and also identifies the oxidizing agent.
← Didn't Know|Knew It →
Consider the following oxidation-reduction reaction.

Which of the following statements is true?
Consider the following oxidation-reduction reaction.
Which of the following statements is true?
Tap to reveal answer
In the reaction, solid potassium (K) is initially in elemental form, so it has an oxidation state of 0. The potassium ions (K+) have a charge of +1 in the product. Since the potassium has lost an electron, it has been oxidized.
Since each hydrogen in reactants (NH3) has an oxidation state of +1, and the hydrogen gas (H2) has a charge of 0 as a product, the hydrogen has been reduced. Since it was reduced, we conclude that NH3 is the oxidizing reagent for the reaction.
In the reaction, solid potassium (K) is initially in elemental form, so it has an oxidation state of 0. The potassium ions (K+) have a charge of +1 in the product. Since the potassium has lost an electron, it has been oxidized.
Since each hydrogen in reactants (NH3) has an oxidation state of +1, and the hydrogen gas (H2) has a charge of 0 as a product, the hydrogen has been reduced. Since it was reduced, we conclude that NH3 is the oxidizing reagent for the reaction.
← Didn't Know|Knew It →
Which compound is oxidized in the following reaction?

Which compound is oxidized in the following reaction?
Tap to reveal answer
Oxidation is defined by a loss of electrons, generally resulting in a more positive charge on an atom. Reduction is defined by a gain of electrons, generally resulting in a more negative charge on an atom. To identify the compound being oxidized, we need to find which compound is becoming more positive.

Zinc is initially neutral, but gains a positive two charge as a product. Hydrogen initially has one positive charge, but becomes neutral in the product. The distinction becomes even clearer if we break the overall reaction into two half-reactions.


We can see that zinc is losing electrons and hydrogen is gaining electrons. Zinc is thus being oxidized.
Oxidation is defined by a loss of electrons, generally resulting in a more positive charge on an atom. Reduction is defined by a gain of electrons, generally resulting in a more negative charge on an atom. To identify the compound being oxidized, we need to find which compound is becoming more positive.
Zinc is initially neutral, but gains a positive two charge as a product. Hydrogen initially has one positive charge, but becomes neutral in the product. The distinction becomes even clearer if we break the overall reaction into two half-reactions.
We can see that zinc is losing electrons and hydrogen is gaining electrons. Zinc is thus being oxidized.
← Didn't Know|Knew It →
Consider the combustion of cyclohexane in air at 298K to give gaseous carbon dioxide and liquid water, as shown in this reaction.

In this reaction, is the oxidizing agent and is the reducing agent.
Consider the combustion of cyclohexane in air at 298K to give gaseous carbon dioxide and liquid water, as shown in this reaction.
In this reaction, is the oxidizing agent and is the reducing agent.
Tap to reveal answer
In this reaction, the oxidation number of oxygen goes from  , in diatomic gaseous oxygen, to
, in diatomic gaseous oxygen, to  , in both carbon dioxide and water. This indicates that it has gained electrons; a gain of electrons indicates that oxygen has been reduced. Since it is reduced, it is the oxidizing agent.
, in both carbon dioxide and water. This indicates that it has gained electrons; a gain of electrons indicates that oxygen has been reduced. Since it is reduced, it is the oxidizing agent.
The oxidation number of carbon goes from  in cyclohexane to
in cyclohexane to  in carbon dioxide. This indicates that it has lost electrons; a loss of electrons indicates that carbon has been oxidized. Since it is oxidized, cyclohexane is the reducing agent.
in carbon dioxide. This indicates that it has lost electrons; a loss of electrons indicates that carbon has been oxidized. Since it is oxidized, cyclohexane is the reducing agent.
In this reaction, the oxidation number of oxygen goes from 

The oxidation number of carbon goes from 

← Didn't Know|Knew It →
Which of the following statements between HClO4 and HClO3 is true?
Which of the following statements between HClO4 and HClO3 is true?
Tap to reveal answer
Oxidation number is a concept that you will also encounter in electrochemistry lectures. It is used to track electrons in an oxidation-reduction reaction. Overall, the sum of the oxidation states of all of the atoms in the molecule must equal the overall charge on the molecule. It is important to remember some of the commonly accepted oxidation states for atoms, so that you can predict the oxidation numbers on other atoms. Here are some key oxidation numbers useful to know for the MCAT:
1. Atoms in elemental form have an oxidation state of 0.
2. Halogens are commonly given an oxidation state of –1.
3. Hydrogen and alkali metals are commonly given an oxidation state of +1.
4. Oxygen is commonly given an oxidation number of –2.
Keeping these rules in mind, we can assume that the hydrogens and oxygens in the two compounds maintain the same oxidation state. Chlorine, however, is attached to one less oxygen in HClO3. As a result, the oxidation state of Cl has decreased from +7 to +5, when comparing HClO4 to HClO3.
Oxidation number is a concept that you will also encounter in electrochemistry lectures. It is used to track electrons in an oxidation-reduction reaction. Overall, the sum of the oxidation states of all of the atoms in the molecule must equal the overall charge on the molecule. It is important to remember some of the commonly accepted oxidation states for atoms, so that you can predict the oxidation numbers on other atoms. Here are some key oxidation numbers useful to know for the MCAT:
1. Atoms in elemental form have an oxidation state of 0.
2. Halogens are commonly given an oxidation state of –1.
3. Hydrogen and alkali metals are commonly given an oxidation state of +1.
4. Oxygen is commonly given an oxidation number of –2.
Keeping these rules in mind, we can assume that the hydrogens and oxygens in the two compounds maintain the same oxidation state. Chlorine, however, is attached to one less oxygen in HClO3. As a result, the oxidation state of Cl has decreased from +7 to +5, when comparing HClO4 to HClO3.
← Didn't Know|Knew It →
Consider the redox reaction:
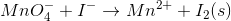
What is the balanced equation for the above redox reaction in acidic solution?
Consider the redox reaction:
What is the balanced equation for the above redox reaction in acidic solution?
Tap to reveal answer

1. Determine the oxidation number of each element.
Reactants: 
Products: 
2. Write and balance the half reactions.
Oxidation (loss of electrons): 
Reduction (gain of electrons): 
3. Balance oxygen by adding water (from the solution) into the half reactions.
Oxidation: 
Reduction: 
4. Balance hydrogen by adding  ions from the acid into the half reactions.
ions from the acid into the half reactions.
Oxidation: 
Reduction: 
5. Multiply each half reaction times an integer such that the electrons cancel when the equations are added. The oxidation reaction will be multiplied by five, and the reduction reaction will be multiplied by two.
Oxidation times 5: 
Reduction times 2: 
6. Add the half reactions together. The electrons will cancel from both the reactants and products.

1. Determine the oxidation number of each element.
Reactants:
Products:
2. Write and balance the half reactions.
Oxidation (loss of electrons):
Reduction (gain of electrons):
3. Balance oxygen by adding water (from the solution) into the half reactions.
Oxidation:
Reduction:
4. Balance hydrogen by adding 
Oxidation:
Reduction:
5. Multiply each half reaction times an integer such that the electrons cancel when the equations are added. The oxidation reaction will be multiplied by five, and the reduction reaction will be multiplied by two.
Oxidation times 5:
Reduction times 2:
6. Add the half reactions together. The electrons will cancel from both the reactants and products.
← Didn't Know|Knew It →
What is the oxidation state of sulfur in H2SO4?
What is the oxidation state of sulfur in H2SO4?
Tap to reveal answer
Since there is no overall charge on the compound the oxidation states must cancel out. Although the oxidation state of hydrides are +1, there are two in the compound that must be accounted for. The same is true for oxygen; although the oxidation number of oxygen is -2, there are four oxygens present accounting for a total of -8. The +2 state contribution from the hydrides and -8 from the oxygens results in a -6 charge. The oxidation state on sulfur must be +6 for the molecule to be neutral.
Since there is no overall charge on the compound the oxidation states must cancel out. Although the oxidation state of hydrides are +1, there are two in the compound that must be accounted for. The same is true for oxygen; although the oxidation number of oxygen is -2, there are four oxygens present accounting for a total of -8. The +2 state contribution from the hydrides and -8 from the oxygens results in a -6 charge. The oxidation state on sulfur must be +6 for the molecule to be neutral.
← Didn't Know|Knew It →
What is the oxidation number of chromium in calcium dichromate (CaCr2O7)?
What is the oxidation number of chromium in calcium dichromate (CaCr2O7)?
Tap to reveal answer
Since the calcium ion has a charge of +2, the dichromate anion must have a charge of -2. Oxygen always has an oxidation number of -2, so the seven oxygens in the molecule have a total number of -14. That leaves a charge of +12 that the two chromiums must balance out (-2 - (-14) = 12). Each chromium must therefore have an oxidation number of +6.
Since the calcium ion has a charge of +2, the dichromate anion must have a charge of -2. Oxygen always has an oxidation number of -2, so the seven oxygens in the molecule have a total number of -14. That leaves a charge of +12 that the two chromiums must balance out (-2 - (-14) = 12). Each chromium must therefore have an oxidation number of +6.
← Didn't Know|Knew It →
Which of the following is an incorrect example of a combustion reaction?
Which of the following is an incorrect example of a combustion reaction?
Tap to reveal answer
We often hear of combustion reactions producing carbon dioxide and water. This is true when the reactants contain all three elements (hydrogen, oxygen, and carbon) needed to make carbon dioxide and water. The trick here is to understand that although combustion is just an oxidation reaction (ie, creating more carbon-oxygen bonds), all reactions must still be balanced. One answer choice fails to balance hydrogen in the reaction.

Although we do see an oxidation reaction with the formation of carbon dioxide, the reaction is NOT balanced. This reaction cannot possibly take place as written.
We often hear of combustion reactions producing carbon dioxide and water. This is true when the reactants contain all three elements (hydrogen, oxygen, and carbon) needed to make carbon dioxide and water. The trick here is to understand that although combustion is just an oxidation reaction (ie, creating more carbon-oxygen bonds), all reactions must still be balanced. One answer choice fails to balance hydrogen in the reaction.
Although we do see an oxidation reaction with the formation of carbon dioxide, the reaction is NOT balanced. This reaction cannot possibly take place as written.
← Didn't Know|Knew It →
The Haber-Bosch process, or simply the Haber process, is a common industrial reaction that generates ammonia from nitrogen and hydrogen gas. A worker in a company generates ammonia from the Haber process. He then dissociates the gaseous ammonia in water to produce an aqueous solution. Since ammonia is a base, it will accept a proton from water, generating  and ammonium ion products. The two reactions involved are:
and ammonium ion products. The two reactions involved are:


A redox (reduction and oxidation) reaction involves a change in the oxidation state of atoms. Oxidation of an atom involves an increase in the oxidation state, whereas reduction involves a decrease in the oxidation state. A oxidized atom will usually become .
The Haber-Bosch process, or simply the Haber process, is a common industrial reaction that generates ammonia from nitrogen and hydrogen gas. A worker in a company generates ammonia from the Haber process. He then dissociates the gaseous ammonia in water to produce an aqueous solution. Since ammonia is a base, it will accept a proton from water, generating 
A redox (reduction and oxidation) reaction involves a change in the oxidation state of atoms. Oxidation of an atom involves an increase in the oxidation state, whereas reduction involves a decrease in the oxidation state. A oxidized atom will usually become .
Tap to reveal answer
The question states that an oxidized atom will increase its oxidation state, which means that the atom will lose electrons and become more positively charged. Since the atom will lose electrons, the number of electrons will be less than the number of protons. Electrons are negatively charged and protons are positively charged; therefore, the presence of more protons in an oxidized atom will make the atom more positive. An oxidized atom will usually become a cation because its number of electrons will decrease.
Reduced atoms will gain electrons and become more negatively charged (decrease oxidation state). Notice that the reactions given in the passage are not redox reactions because the atoms in both reactions retain their oxidation states.
The question states that an oxidized atom will increase its oxidation state, which means that the atom will lose electrons and become more positively charged. Since the atom will lose electrons, the number of electrons will be less than the number of protons. Electrons are negatively charged and protons are positively charged; therefore, the presence of more protons in an oxidized atom will make the atom more positive. An oxidized atom will usually become a cation because its number of electrons will decrease.
Reduced atoms will gain electrons and become more negatively charged (decrease oxidation state). Notice that the reactions given in the passage are not redox reactions because the atoms in both reactions retain their oxidation states.
← Didn't Know|Knew It →
What is the oxidation state of nitrogen in HNO3?
What is the oxidation state of nitrogen in HNO3?
Tap to reveal answer
When applying oxidation numbers there are certain hierarchical rules that must be followed.
1. The sum of oxidation states of all the elements in a molecule must add up to the overall charge.
2. Group 1 and Group 2 elements have +1 and +2 oxidation states, respectively.
3. Fluorine has an oxidation state of –1.
4. Hydrogen has an oxidation state of +1 (except in metal hydrides).
5. Oxygen has an oxidation state of –2.
6. Elements of the same group (excluding transition metals) generally have the same oxidation state.
When applying the oxidation state to HNO3, hydrogen has a +1 oxidation state, and each of the oxygen molecules has a –2 oxidation state. Since there is only one hydrogen molecule and three oxygen molecules, the oxidation state of nitrogen must balance out the charge of the hydrogen molecule and oxygen molecules combined.



Nitrogen must have a +5 oxidation state.
When applying oxidation numbers there are certain hierarchical rules that must be followed.
1. The sum of oxidation states of all the elements in a molecule must add up to the overall charge.
2. Group 1 and Group 2 elements have +1 and +2 oxidation states, respectively.
3. Fluorine has an oxidation state of –1.
4. Hydrogen has an oxidation state of +1 (except in metal hydrides).
5. Oxygen has an oxidation state of –2.
6. Elements of the same group (excluding transition metals) generally have the same oxidation state.
When applying the oxidation state to HNO3, hydrogen has a +1 oxidation state, and each of the oxygen molecules has a –2 oxidation state. Since there is only one hydrogen molecule and three oxygen molecules, the oxidation state of nitrogen must balance out the charge of the hydrogen molecule and oxygen molecules combined.
Nitrogen must have a +5 oxidation state.
← Didn't Know|Knew It →
What is the oxidation number of hydrogen in NaH?
What is the oxidation number of hydrogen in NaH?
Tap to reveal answer
When applying oxidation numbers there are certain hierarchical rules that must be followed.
1. The sum of oxidation states of all the elements in a molecule must add up to the overall charge.
2. Group 1 and Group 2 elements have +1 and +2 oxidation states, respectively.
3. Fluorine has an oxidation state of –1.
4. Hydrogen has an oxidation state of +1 (except in metal hydrides).
5. Oxygen has an oxidation state of –2.
6. Elements of the same group (excluding transition metals) generally have the same oxidation state.
Following the oxidation rules, group 1 elements must have an oxidation number of +1.


To balance out the overall charge of zero, the hydrogen in this compound must have an oxidation state of –1. Note that NaH is a metal hydride; therefore, the oxidation state of hydrogen is not necessarily +1.
When applying oxidation numbers there are certain hierarchical rules that must be followed.
1. The sum of oxidation states of all the elements in a molecule must add up to the overall charge.
2. Group 1 and Group 2 elements have +1 and +2 oxidation states, respectively.
3. Fluorine has an oxidation state of –1.
4. Hydrogen has an oxidation state of +1 (except in metal hydrides).
5. Oxygen has an oxidation state of –2.
6. Elements of the same group (excluding transition metals) generally have the same oxidation state.
Following the oxidation rules, group 1 elements must have an oxidation number of +1.
To balance out the overall charge of zero, the hydrogen in this compound must have an oxidation state of –1. Note that NaH is a metal hydride; therefore, the oxidation state of hydrogen is not necessarily +1.
← Didn't Know|Knew It →
Boiling point is the temperature a liquid needs to achieve in order to begin its transformation into a gaseous state. Campers and hikers who prepare food during their trips have to account for differences in atmospheric pressure as they ascend in elevation. During the ascent, the decrease in atmospheric pressure changes the temperature at which water boils.
Further complicating the matter is the observation that addition of a solute to a pure liquid also changes the boiling point. Raoult’s Law can be used to understand the changes in boiling point if a non-volatile solute is present, as expressed here.

In this law,  is the mole fraction of the solvent,
is the mole fraction of the solvent,  is the vapor pressure of the pure solvent, and
is the vapor pressure of the pure solvent, and  is the vapor pressure of the solution. When this vapor pressure is equal to the local atmospheric pressure, the solution boils.
is the vapor pressure of the solution. When this vapor pressure is equal to the local atmospheric pressure, the solution boils.
The change in boiling point with addition of a solute is a colligative property of a solution. Which of the following are also examples of colligative properties?
I. Vapor pressure reduction
II. Color emission with dissolution of a solute
III. Osmotic pressure
Boiling point is the temperature a liquid needs to achieve in order to begin its transformation into a gaseous state. Campers and hikers who prepare food during their trips have to account for differences in atmospheric pressure as they ascend in elevation. During the ascent, the decrease in atmospheric pressure changes the temperature at which water boils.
Further complicating the matter is the observation that addition of a solute to a pure liquid also changes the boiling point. Raoult’s Law can be used to understand the changes in boiling point if a non-volatile solute is present, as expressed here.
In this law, 


The change in boiling point with addition of a solute is a colligative property of a solution. Which of the following are also examples of colligative properties?
I. Vapor pressure reduction
II. Color emission with dissolution of a solute
III. Osmotic pressure
Tap to reveal answer
Colligative properties are defined as properties that depend entirely upon the ratio of the number of solute particles to the number of solvent particles. Only osmotic pressure and vapor pressure depression are examples of such phenomena. While color emission is a property of a solution, it depends on the chemical species involved, and not the number of particles.
Colligative properties are defined as properties that depend entirely upon the ratio of the number of solute particles to the number of solvent particles. Only osmotic pressure and vapor pressure depression are examples of such phenomena. While color emission is a property of a solution, it depends on the chemical species involved, and not the number of particles.
← Didn't Know|Knew It →
Boiling point is the temperature a liquid needs to achieve in order to begin its transformation into a gaseous state. Campers and hikers who prepare food during their trips have to account for differences in atmospheric pressure as they ascend in elevation. During the ascent, the decrease in atmospheric pressure changes the temperature at which water boils.
Further complicating the matter is the observation that addition of a solute to a pure liquid also changes the boiling point. Raoult’s Law can be used to understand the changes in boiling point if a non-volatile solute is present, as expressed here.

In this law,  is the mole fraction of the solvent,
is the mole fraction of the solvent,  is the vapor pressure of the pure solvent, and
is the vapor pressure of the pure solvent, and  is the vapor pressure of the solution. When this vapor pressure is equal to the local atmospheric pressure, the solution boils.
is the vapor pressure of the solution. When this vapor pressure is equal to the local atmospheric pressure, the solution boils.
Two campers are preparing food at an altitude of 13,000 feet on a mountain in Colorado. Which of the following is true as they boil a pot of water?
Boiling point is the temperature a liquid needs to achieve in order to begin its transformation into a gaseous state. Campers and hikers who prepare food during their trips have to account for differences in atmospheric pressure as they ascend in elevation. During the ascent, the decrease in atmospheric pressure changes the temperature at which water boils.
Further complicating the matter is the observation that addition of a solute to a pure liquid also changes the boiling point. Raoult’s Law can be used to understand the changes in boiling point if a non-volatile solute is present, as expressed here.
In this law, 


Two campers are preparing food at an altitude of 13,000 feet on a mountain in Colorado. Which of the following is true as they boil a pot of water?
Tap to reveal answer
The local atmospheric pressure at 13,000 feet is less than the pressure at sea level; therefore, it takes less heat to make the vapor pressure meet the local atmospheric pressure. Heat added to the system easily exits again as the water is converted to steam, leaving less heat in the water to cook the food. Food cooks more slowly as a result.
The local atmospheric pressure at 13,000 feet is less than the pressure at sea level; therefore, it takes less heat to make the vapor pressure meet the local atmospheric pressure. Heat added to the system easily exits again as the water is converted to steam, leaving less heat in the water to cook the food. Food cooks more slowly as a result.
← Didn't Know|Knew It →
Which solution will have a higher boiling point?
Solution 1:  in
in  water
water
Solution 2:  in
in  water
water
Which solution will have a higher boiling point?
Solution 1: 

Solution 2: 

Tap to reveal answer
Adding solute to water will result in boiling point elevation due to the presence of more molecules. Change in temperature is given by the relation  , where
, where  is a constant for the solvent,
is a constant for the solvent,  is the solution molality, and
is the solution molality, and  is the van't Hoff factor. In this example, the molalities are equal.
is the van't Hoff factor. In this example, the molalities are equal.
Since  dissociates into
dissociates into  and
and  ,
,  , representing the two ions derived from each molecule. For glucose
, representing the two ions derived from each molecule. For glucose  , as the molecule does not dissociate. Solution 1 will have a higher elevation in temperature due to the greater number of ions in solution.
, as the molecule does not dissociate. Solution 1 will have a higher elevation in temperature due to the greater number of ions in solution.
Adding solute to water will result in boiling point elevation due to the presence of more molecules. Change in temperature is given by the relation 



Since 




← Didn't Know|Knew It →
Compound Base strength, K b 1 1012 2 105 3 101 4 10-8
Students in a chemistry class are given one of four unknown samples in a laboratory. A student is told that his compound is the strongest acid of the four compounds. Based on the information in the above table, which compound was the student given?
| Compound | Base strength, K b |
|---|---|
| 1 | 1012 |
| 2 | 105 |
| 3 | 101 |
| 4 | 10-8 |
Students in a chemistry class are given one of four unknown samples in a laboratory. A student is told that his compound is the strongest acid of the four compounds. Based on the information in the above table, which compound was the student given?
Tap to reveal answer
The strongest acid of the group will also have the smallest Kb value. As the weakest base (smallest Kb), compound 4 will only partially dissociate in solution because it has a fairly strong conjugate acid. Were this question asking which base was the strongest, compound 1 would be the answer, due to its large Kb value.
The strongest acid of the group will also have the smallest Kb value. As the weakest base (smallest Kb), compound 4 will only partially dissociate in solution because it has a fairly strong conjugate acid. Were this question asking which base was the strongest, compound 1 would be the answer, due to its large Kb value.
← Didn't Know|Knew It →
Each of the following solutions is added to equal amounts of water. Which solution will result in the greatest amount of boiling point elevation?
Each of the following solutions is added to equal amounts of water. Which solution will result in the greatest amount of boiling point elevation?
Tap to reveal answer
Boiling point elevation is a colligative property, meaning that it depends on the relative number of solute particles in solution. The answer choice with the largest number of moles of particles will show the greatest boiling point elevation. The equation for boiling point elevation is:

Molality is equal to moles of solute per kilogram of solvent, meaning that it will be proportional to the moles of solute added. Each solute is added to equal amounts of water, allowing us to keep this value constant. Similarly,  will be constant for all of the solutions. Overall, boiling point elevation will be proportional to the moles of solute multiplied by the van't Hoff factor.
will be constant for all of the solutions. Overall, boiling point elevation will be proportional to the moles of solute multiplied by the van't Hoff factor.

Using this proportion, we can find the solute that will most impact the boiling point of water.





Since sodium chloride results in the greatest moles of ions in solution, it will yield the greatest boiling point elevation.
Boiling point elevation is a colligative property, meaning that it depends on the relative number of solute particles in solution. The answer choice with the largest number of moles of particles will show the greatest boiling point elevation. The equation for boiling point elevation is:
Molality is equal to moles of solute per kilogram of solvent, meaning that it will be proportional to the moles of solute added. Each solute is added to equal amounts of water, allowing us to keep this value constant. Similarly, 
Using this proportion, we can find the solute that will most impact the boiling point of water.
Since sodium chloride results in the greatest moles of ions in solution, it will yield the greatest boiling point elevation.
← Didn't Know|Knew It →
What is the correct oxidation number of zinc in the compound  ?
?
What is the correct oxidation number of zinc in the compound 
Tap to reveal answer
The sum of oxidation numbers of each element in this compound must add to the total charge of -2. Hydrogen always has an oxidation number of +1, and oxygen always has an oxidation number of -2.
So, in total for oxygen we have (4) * (-2) = -8, and for hydrogen we have (4) * (+1) = +4.
Combining the oxygen and hydrogen, (+4) + (-8) = -4, so we need an additional +2 to achieve the total charge of -2. Zn must provide this balancing charge, and have an oxidation number of +2.
The sum of oxidation numbers of each element in this compound must add to the total charge of -2. Hydrogen always has an oxidation number of +1, and oxygen always has an oxidation number of -2.
So, in total for oxygen we have (4) * (-2) = -8, and for hydrogen we have (4) * (+1) = +4.
Combining the oxygen and hydrogen, (+4) + (-8) = -4, so we need an additional +2 to achieve the total charge of -2. Zn must provide this balancing charge, and have an oxidation number of +2.
← Didn't Know|Knew It →
What is the molarity of a 1L solution composed of water and 300g of sodium iodide?
What is the molarity of a 1L solution composed of water and 300g of sodium iodide?
Tap to reveal answer
Sodium iodide is given by the formula NaI, and has a molecular weight of 150g/mol. Molarity is found by dividing the moles of solute by liters of solvent.

We find the moles of sodium iodide by using the mass (300g) and molecular weight.

We know our volume is 1L, so now we can solve for the molarity.

Sodium iodide is given by the formula NaI, and has a molecular weight of 150g/mol. Molarity is found by dividing the moles of solute by liters of solvent.
We find the moles of sodium iodide by using the mass (300g) and molecular weight.
We know our volume is 1L, so now we can solve for the molarity.
← Didn't Know|Knew It →
All of the following are true regarding a solution except .
All of the following are true regarding a solution except .
Tap to reveal answer
Solutions can occur in all three phases of matter. They can also occur between two compounds in a single phase. For example, brass is a solution of two metals: zinc and copper.
Solutions can occur in all three phases of matter. They can also occur between two compounds in a single phase. For example, brass is a solution of two metals: zinc and copper.
← Didn't Know|Knew It →