Thermochemistry and Kinetics - AP Chemistry
Card 1 of 2017

Suppose that 1.5 moles of potassium reacts completely with an excess of chlorine gas. What is the enthalpy change of the reaction?
Suppose that 1.5 moles of potassium reacts completely with an excess of chlorine gas. What is the enthalpy change of the reaction?
Tap to reveal answer
First, note that heat is a product of the reaction. This means that the reaction releases heat and is exothermic, so the enthalpy change must be negative. Next, consider that 2 moles of sodium would react to produce 640 kJ. Thus, 1.5 moles of sodium would react to produce
 .
.
Hence, the enthalpy change is 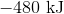 .
.
First, note that heat is a product of the reaction. This means that the reaction releases heat and is exothermic, so the enthalpy change must be negative. Next, consider that 2 moles of sodium would react to produce 640 kJ. Thus, 1.5 moles of sodium would react to produce

Hence, the enthalpy change is 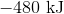
← Didn't Know|Knew It →
The second law of thermodynamics states which of the following is true regarding an isolated system?
The second law of thermodynamics states which of the following is true regarding an isolated system?
Tap to reveal answer
The entropy cannot decrease in an isolated system because the energy can only be degraded. Since the system is isolated, no higher-grade energy—or any energy at all—is being introduced into the system. As a result, the entropy cannot decrease. The other answer choices relate to the other laws of thermodynamics.
The entropy cannot decrease in an isolated system because the energy can only be degraded. Since the system is isolated, no higher-grade energy—or any energy at all—is being introduced into the system. As a result, the entropy cannot decrease. The other answer choices relate to the other laws of thermodynamics.
← Didn't Know|Knew It →
Which of the following statements is true of standard states?
Which of the following statements is true of standard states?
Tap to reveal answer
Standard states are defined as a specific set of conditions, such as when a gas is at  ,
,  concentration, and
concentration, and  .
.
Standard enthalpy of formation, the energy required for form 1 mole of a compound from its constituent elements, occurs when elements are in their standard states.
Standard states are defined as a specific set of conditions, such as when a gas is at 


Standard enthalpy of formation, the energy required for form 1 mole of a compound from its constituent elements, occurs when elements are in their standard states.
← Didn't Know|Knew It →
How much heat is required to raise the temperature of  of water from
of water from  to
to  ? (Specific heat capacity of water is
? (Specific heat capacity of water is  )
)
How much heat is required to raise the temperature of 



Tap to reveal answer

 is positive because heat flows into the system to raise the temperature of the water.
is positive because heat flows into the system to raise the temperature of the water.

← Didn't Know|Knew It →
How much heat is required to raise the temperature of  of water from
of water from  to
to  ? Specific heat capacity of water is
? Specific heat capacity of water is 
How much heat is required to raise the temperature of 


Tap to reveal answer
← Didn't Know|Knew It →
Calculating heat
How much heat is absorbed by a copper penny as it warms from  to
to  assuming the penny is pure copper with a mass of
assuming the penny is pure copper with a mass of  ?
?  of copper is
of copper is  .
.
Calculating heat
How much heat is absorbed by a copper penny as it warms from 




Tap to reveal answer
Use the equation that relates heat, mass, specific heat, and change in temperature:



Use the equation that relates heat, mass, specific heat, and change in temperature:
← Didn't Know|Knew It →
"In a natural thermodynamic process, the sum of the entropies of the interacting systems increases." Which law of thermodynamics does this statement refer to?
"In a natural thermodynamic process, the sum of the entropies of the interacting systems increases." Which law of thermodynamics does this statement refer to?
Tap to reveal answer
There are four main laws of thermodynamics, which describe how temperature, energy, and entropy behave under various circumstances. The zeroth law of thermodynamics helps to define temperature; it states that if two systems are each in thermal equilibrium with a third system, they must be in thermal equilibrium with each other. The first law of thermodynamics negates the possibility of perpetual motion; it states that when energy passes into or out of a system, the system's internal energy changes in accord with the law of conservation of energy. The second law of thermodynamics also negates the possibility of perpetual motion; it states that in a natural thermodynamic process, the sum of the entropies of the interacting systems increases. Lastly, the third law of thermodynamics states that the entropy of a system approaches a constant value as the temperature nears absolute zero.
There are four main laws of thermodynamics, which describe how temperature, energy, and entropy behave under various circumstances. The zeroth law of thermodynamics helps to define temperature; it states that if two systems are each in thermal equilibrium with a third system, they must be in thermal equilibrium with each other. The first law of thermodynamics negates the possibility of perpetual motion; it states that when energy passes into or out of a system, the system's internal energy changes in accord with the law of conservation of energy. The second law of thermodynamics also negates the possibility of perpetual motion; it states that in a natural thermodynamic process, the sum of the entropies of the interacting systems increases. Lastly, the third law of thermodynamics states that the entropy of a system approaches a constant value as the temperature nears absolute zero.
← Didn't Know|Knew It →
The following reversible reaction takes place in a closed container:

The system is allowed to reach equilibrium before 2 moles of reactant A are added to the container.
The net rate of formation of product B immediately following the addition of reactant A is .
The following reversible reaction takes place in a closed container:
The system is allowed to reach equilibrium before 2 moles of reactant A are added to the container.
The net rate of formation of product B immediately following the addition of reactant A is .
Tap to reveal answer
According to Le Chatelier's principle, an increase in the concentration of reactants will cause the reaction to shift to the right in order to re-establish equilibrium. Thus, immediately after reactant A is added, the forward reaction will increase and the net rate of formation of the products will be positive.
According to Le Chatelier's principle, an increase in the concentration of reactants will cause the reaction to shift to the right in order to re-establish equilibrium. Thus, immediately after reactant A is added, the forward reaction will increase and the net rate of formation of the products will be positive.
← Didn't Know|Knew It →
Rate data is collected for a reaction, and the following integrated rate law is derived.
![ln[A] = ln[A_0] - kt](https://vt-vtwa-assets.varsitytutors.com/vt-vtwa/uploads/formula_image/image/335812/gif.latex)
If this reaction is with respect to reactant A, a plot of ln\[A\] versus time would be .
Rate data is collected for a reaction, and the following integrated rate law is derived.
If this reaction is with respect to reactant A, a plot of ln\[A\] versus time would be .
Tap to reveal answer
The integrated rate law shown is for a reaction rate law with a first-order dependence on reactact A. If such a rate law accurately describes the kinetics of the reaction, then ln\[A\] will vary linearly with respect to time.
The integrated rate law shown is for a reaction rate law with a first-order dependence on reactact A. If such a rate law accurately describes the kinetics of the reaction, then ln\[A\] will vary linearly with respect to time.
← Didn't Know|Knew It →
Which of the following affects the rate of a reaction?
Which of the following affects the rate of a reaction?
Tap to reveal answer
Since reactant molecules collide and interact to break old bond and form new ones, any factors affecting collision and interactions will affect the reaction rate. Thus, increasing temperature will increase both the frequency of collisions and the average kinetic energy of the molecules. Enzymes bring molecules close to each other and orient them in a way that facilitates reactions.
Since reactant molecules collide and interact to break old bond and form new ones, any factors affecting collision and interactions will affect the reaction rate. Thus, increasing temperature will increase both the frequency of collisions and the average kinetic energy of the molecules. Enzymes bring molecules close to each other and orient them in a way that facilitates reactions.
← Didn't Know|Knew It →
If the activation energy of a forward reaction is greater than the activation energy of a reverse reaction, what must be true of the reaction?
If the activation energy of a forward reaction is greater than the activation energy of a reverse reaction, what must be true of the reaction?
Tap to reveal answer
If the activation energy of the forward reaction is greater than that of the reverse reaction, the products must have a higher enthalpy than the reactants. The net enthalpy change is therefore positive, meaning that it is endothermic.
If the activation energy of the forward reaction is greater than that of the reverse reaction, the products must have a higher enthalpy than the reactants. The net enthalpy change is therefore positive, meaning that it is endothermic.
← Didn't Know|Knew It →
If the activation energy of the forward reaction is greater than the activatoin energy of the reverse reaction, then this reaction is .
If the activation energy of the forward reaction is greater than the activatoin energy of the reverse reaction, then this reaction is .
Tap to reveal answer
If the activation energy of a forward reaction is greater than that of the reverse reaction, thenthe products must have a higher enthalpy than the reactants (draw a potential energy diagram to visualize).
If the activation energy of a forward reaction is greater than that of the reverse reaction, thenthe products must have a higher enthalpy than the reactants (draw a potential energy diagram to visualize).
← Didn't Know|Knew It →
The overall reaction can only proceed as quickly as the .
The overall reaction can only proceed as quickly as the .
Tap to reveal answer
The rate-determining step in a reaction mechanism is a kinetic bottleneck, in that it prevents the overall reaction from proceeding; thus, it is what determines how quickly the overall reaction can proceed.
The rate-determining step in a reaction mechanism is a kinetic bottleneck, in that it prevents the overall reaction from proceeding; thus, it is what determines how quickly the overall reaction can proceed.
← Didn't Know|Knew It →
Calculate the standard cell potential of the following reaction:
Zn (s) + 2 Ag1+ (aq) -> Zn2+ (aq) + 2 Ag (s)
Given:
Zn2+(aq)+ 2 e--> Zn (s) Eo = -0.76 V
Ag1+(aq)+ 1 e--> Ag (s) Eo = 0.80 V
Calculate the standard cell potential of the following reaction:
Zn (s) + 2 Ag1+ (aq) -> Zn2+ (aq) + 2 Ag (s)
Given:
Zn2+(aq)+ 2 e--> Zn (s) Eo = -0.76 V
Ag1+(aq)+ 1 e--> Ag (s) Eo = 0.80 V
Tap to reveal answer
Eocell = Eo cathode - Eoanode
Eocell = 0.80 – (-0.76) = 1.56 V
Eocell = Eo cathode - Eoanode
Eocell = 0.80 – (-0.76) = 1.56 V
← Didn't Know|Knew It →
Calculate the standard cell potential of the following reaction:
3 F2 (g) + 2 Au (s) -> 6 F- (aq) + 2 Au3+
Given:
F2 (g) + 2 e- -> 2 F- (aq) Eo = 2.87 V
Au3+(aq)+ 3 e--> Au (s) Eo = 1.50 V
Calculate the standard cell potential of the following reaction:
3 F2 (g) + 2 Au (s) -> 6 F- (aq) + 2 Au3+
Given:
F2 (g) + 2 e- -> 2 F- (aq) Eo = 2.87 V
Au3+(aq)+ 3 e--> Au (s) Eo = 1.50 V
Tap to reveal answer
Eocell = Eo cathode - Eoanode
Eocell = 2.87 – (1.50) = 1.37 V
Eocell = Eo cathode - Eoanode
Eocell = 2.87 – (1.50) = 1.37 V
← Didn't Know|Knew It →
Which of the following is true regarding activation energy?
I. Activation energy depends on the pressure of the system
II. The reaction rate decreases as the activation energy increases
III. Catalysts increase the activation energy of a reaction
Which of the following is true regarding activation energy?
I. Activation energy depends on the pressure of the system
II. The reaction rate decreases as the activation energy increases
III. Catalysts increase the activation energy of a reaction
Tap to reveal answer
Activation energy is the energy barrier that needs to be overcome for a reaction to proceed; the higher the activation energy, the slower the reaction. Activation energy can only be altered via a catalyst. Catalysts are chemical substances that lower the activation energy, allowing reactions to proceed faster. Other physical quantities such as temperature and pressure don’t alter the activation energy.
Reaction rate is a direct measure of the speed of a reaction. As mentioned, increasing activation energy will increase the barrier and, therefore, slow down the reaction.
Catalysts are chemical substances that decrease the activation energy, thereby increasing the reaction rate. They are commonly used in chemical reactions to drastically speed up reactions that might otherwise take hours or days to complete. Enzymes are biological catalysts that facilitate most of the biological reactions happening in our bodies.
Activation energy is the energy barrier that needs to be overcome for a reaction to proceed; the higher the activation energy, the slower the reaction. Activation energy can only be altered via a catalyst. Catalysts are chemical substances that lower the activation energy, allowing reactions to proceed faster. Other physical quantities such as temperature and pressure don’t alter the activation energy.
Reaction rate is a direct measure of the speed of a reaction. As mentioned, increasing activation energy will increase the barrier and, therefore, slow down the reaction.
Catalysts are chemical substances that decrease the activation energy, thereby increasing the reaction rate. They are commonly used in chemical reactions to drastically speed up reactions that might otherwise take hours or days to complete. Enzymes are biological catalysts that facilitate most of the biological reactions happening in our bodies.
← Didn't Know|Knew It →
the activation energy of a reaction will the amount of products produced.
the activation energy of a reaction will the amount of products produced.
Tap to reveal answer
This question is asking about the relationship between activation energy of a reaction (kinetics) and the amount of products produced (equilibrium). Remember that altering the speed of a reaction (kinetics) does not change the equilibrium of the reaction. Increasing or decreasing activation energy (which alters the speed of reaction) will simply allow for the reaction to proceed slower or faster, respectively. It will not change the amount of products produced at the end.
This question is asking about the relationship between activation energy of a reaction (kinetics) and the amount of products produced (equilibrium). Remember that altering the speed of a reaction (kinetics) does not change the equilibrium of the reaction. Increasing or decreasing activation energy (which alters the speed of reaction) will simply allow for the reaction to proceed slower or faster, respectively. It will not change the amount of products produced at the end.
← Didn't Know|Knew It →
Which of the following is the correct way to calculate the activation energy of a reaction?
Which of the following is the correct way to calculate the activation energy of a reaction?
Tap to reveal answer
Activation energy is the difference between the energy of the transition state and the energy of the reactant. Recall that activation energy is the energy barrier that needs to be overcome by a reaction. The transition state is a higher-energy, intermediary molecule that lies in between the reactants and products. It is created when reactants are in the process of becoming products. Therefore, to get to products, it is necessary to go through this high-energy transition state.
Activation energy is the energy needed to climb this energy “hill” (energy needed to go from reactants to transition state); therefore, the activation energy is the energy of the transition state (top of the energy hill/barrier) minus the energy of the reactant.
Activation energy is the difference between the energy of the transition state and the energy of the reactant. Recall that activation energy is the energy barrier that needs to be overcome by a reaction. The transition state is a higher-energy, intermediary molecule that lies in between the reactants and products. It is created when reactants are in the process of becoming products. Therefore, to get to products, it is necessary to go through this high-energy transition state.
Activation energy is the energy needed to climb this energy “hill” (energy needed to go from reactants to transition state); therefore, the activation energy is the energy of the transition state (top of the energy hill/barrier) minus the energy of the reactant.
← Didn't Know|Knew It →
Which of the following is true of catalysts?
Which of the following is true of catalysts?
Tap to reveal answer
Catalysts are substances that increase reaction rates without being consumed in the reaction. They decrease the activation energy needed, and they do not always need to be in the same phase as the reactants. In heterogeneous catalysis, the catalyst is in a different phase than the reactants. Equilibrium concentrations of both reactants and products are unchanged by the addition of a catalyst.
Catalysts are substances that increase reaction rates without being consumed in the reaction. They decrease the activation energy needed, and they do not always need to be in the same phase as the reactants. In heterogeneous catalysis, the catalyst is in a different phase than the reactants. Equilibrium concentrations of both reactants and products are unchanged by the addition of a catalyst.
← Didn't Know|Knew It →
Which of the following is not true of catalysts?
Which of the following is not true of catalysts?
Tap to reveal answer
All of the choices are true, except that catalysts can be in distinct phases than the reactants. These are known as heterogenous catalysts.
All of the choices are true, except that catalysts can be in distinct phases than the reactants. These are known as heterogenous catalysts.
← Didn't Know|Knew It →

