Laboratory Techniques and Analysis - AP Chemistry
Card 1 of 726
Which choice of lab equipment is never used to make accurate volume measurements?
Which choice of lab equipment is never used to make accurate volume measurements?
Tap to reveal answer
Graduated cylinders are often used for measuring volumetric quantities. As these have several markings, they are considered accurate. Volumetric flasks are also used to accurately measure liquid volume quantities. Lastly, burettes are used to accurately measure volumes of liquid during titrations.
In contrast, beakers are not typically used to measure accurate quantities of liquid; though they have markings on them, these are approximate measurements. Graduated cylinders are often used to accurately measure out a liquid, and beakers to then hold the liquid.
Graduated cylinders are often used for measuring volumetric quantities. As these have several markings, they are considered accurate. Volumetric flasks are also used to accurately measure liquid volume quantities. Lastly, burettes are used to accurately measure volumes of liquid during titrations.
In contrast, beakers are not typically used to measure accurate quantities of liquid; though they have markings on them, these are approximate measurements. Graduated cylinders are often used to accurately measure out a liquid, and beakers to then hold the liquid.
← Didn't Know|Knew It →
The literature value for the melting point of a given compound is 240 degrees Celsius. A scientist uses the melting point apparatus to experimentally measure the compound's melting point at 230 degrees Celsius. What is the scientist's percent error?
The literature value for the melting point of a given compound is 240 degrees Celsius. A scientist uses the melting point apparatus to experimentally measure the compound's melting point at 230 degrees Celsius. What is the scientist's percent error?
Tap to reveal answer
The percent error is given by the following formula:

In this case, the theoretical value is 240 degrees Celsius, and the actual value is 230 degrees Celsius. The percent error is thus calculated to be 4.16 %. The calculation is as follows:

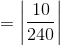


The percent error is given by the following formula:
In this case, the theoretical value is 240 degrees Celsius, and the actual value is 230 degrees Celsius. The percent error is thus calculated to be 4.16 %. The calculation is as follows:
← Didn't Know|Knew It →
An experiment requires you to mix a solution of  with a solution of
with a solution of  and then observe the reaction. What should the solutions be mixed in?
and then observe the reaction. What should the solutions be mixed in?
An experiment requires you to mix a solution of 

Tap to reveal answer
A beaker is the best choice for mixing these two solutions. Graduated cylinders, burets, and pipets are all used to measure specific amounts of solution. Also, stirbars can be easily added to beakers to facilitate the reaction.
A beaker is the best choice for mixing these two solutions. Graduated cylinders, burets, and pipets are all used to measure specific amounts of solution. Also, stirbars can be easily added to beakers to facilitate the reaction.
← Didn't Know|Knew It →
Your lab partner is asking you how to properly label a solution of HCl. Which label did you show him?
Your lab partner is asking you how to properly label a solution of HCl. Which label did you show him?
Tap to reveal answer
When you label chemical solutions, you should technically always use the full chemical name (though this is not always done). Using common abbreviations, like HCl for hydrochloric acid, is generally acceptable.
The reason why none of the other answers work is because they did not quantify how strong (concentrated) the HCl solution was. A 1 molar HCl solution is handled much differently than a 12 molar one. Even though the answer choice "1M of acid" did quantify the concentration, it did not say which acid was in solution, which would be very unsafe if someone else saw your beaker and needed to know how to dispose of the contents.
Labels should always provide information on the contents of the container, as well as its concentration strength.
When you label chemical solutions, you should technically always use the full chemical name (though this is not always done). Using common abbreviations, like HCl for hydrochloric acid, is generally acceptable.
The reason why none of the other answers work is because they did not quantify how strong (concentrated) the HCl solution was. A 1 molar HCl solution is handled much differently than a 12 molar one. Even though the answer choice "1M of acid" did quantify the concentration, it did not say which acid was in solution, which would be very unsafe if someone else saw your beaker and needed to know how to dispose of the contents.
Labels should always provide information on the contents of the container, as well as its concentration strength.
← Didn't Know|Knew It →
You have a stock solution of 12M HCl, but want to dilute it to 1M HCl. What is the safest way you could go about doing this?
You have a stock solution of 12M HCl, but want to dilute it to 1M HCl. What is the safest way you could go about doing this?
Tap to reveal answer
Always add acid to water. You should mix solutions slowly because you run the risk of solutions spilling, which may ruin your reaction entirely. Rapid mixing can also lead to inadvertently speeding up reactions and releasing energy or heat in a dangerous manner. During dilution of acids with, water heat is released; if a small amount of water is added to a concentrated acid it has the possibility of bubbling over the counter and onto you.
Always add acid to water. You should mix solutions slowly because you run the risk of solutions spilling, which may ruin your reaction entirely. Rapid mixing can also lead to inadvertently speeding up reactions and releasing energy or heat in a dangerous manner. During dilution of acids with, water heat is released; if a small amount of water is added to a concentrated acid it has the possibility of bubbling over the counter and onto you.
← Didn't Know|Knew It →
Which procedure separates two substances using their different boiling points?
Which procedure separates two substances using their different boiling points?
Tap to reveal answer
During the distillation process, the combined substances are heated until the first one boils. The substance that boils and becomes a gas is then separated from the still liquid or solid substance via condensation into a separate container. Filtration involves separating substances in solid and liquid phases through a filter paper, and is often facilitated by a vacuum. Dehydration and hydration are types of reactions where water is a product, and a reactant, respectively. Fermentation is a biochemical process that occurs under anaerobic conditions. Some organisms produce lactic acid as a byproduct of fermentation, (humans), others produce ethanol (yeast).
During the distillation process, the combined substances are heated until the first one boils. The substance that boils and becomes a gas is then separated from the still liquid or solid substance via condensation into a separate container. Filtration involves separating substances in solid and liquid phases through a filter paper, and is often facilitated by a vacuum. Dehydration and hydration are types of reactions where water is a product, and a reactant, respectively. Fermentation is a biochemical process that occurs under anaerobic conditions. Some organisms produce lactic acid as a byproduct of fermentation, (humans), others produce ethanol (yeast).
← Didn't Know|Knew It →
You are trying to create a new chemical structure. You need exactly 9mL of phenyllithium for your next reaction. Which measuring tool would be the most accurate to measure out that liquid?
You are trying to create a new chemical structure. You need exactly 9mL of phenyllithium for your next reaction. Which measuring tool would be the most accurate to measure out that liquid?
Tap to reveal answer
When measuring any liquid in the lab, a volumetric pipet or flask is always going to be the most accurate. This makes the 9mL volumetric pipet the best choice.
There is no such thing as a 9mL watch glass. Though a 10mL syringe and graduated cylinder would be quite accurate, the 9mL volumetric pipet is more accurate, and remember the question asked for the best out of the answer choices. A beaker is very inaccurate and should only be used to transfer solutions, not to measure them.
When measuring any liquid in the lab, a volumetric pipet or flask is always going to be the most accurate. This makes the 9mL volumetric pipet the best choice.
There is no such thing as a 9mL watch glass. Though a 10mL syringe and graduated cylinder would be quite accurate, the 9mL volumetric pipet is more accurate, and remember the question asked for the best out of the answer choices. A beaker is very inaccurate and should only be used to transfer solutions, not to measure them.
← Didn't Know|Knew It →
An experiment calls for you to titrate a  solution of
solution of  with a solution of
with a solution of  of unknown concentration. The solution of
of unknown concentration. The solution of  is held in an Erlenmeyer flask. What piece of laboratory equipment should you use to administer the
is held in an Erlenmeyer flask. What piece of laboratory equipment should you use to administer the  solution?
solution?
An experiment calls for you to titrate a 




Tap to reveal answer
In a titration, you should use a buret to administer the  solution into the
solution into the  solution. A buret gives you the most accuracy in running the titration as you can dispense the solution drops at a time. The buret will also allow you to accurately determine the volume of
solution. A buret gives you the most accuracy in running the titration as you can dispense the solution drops at a time. The buret will also allow you to accurately determine the volume of  needed to complete the titration. A watch glass and spatula would be inappropriate for this situation because you are dealing with liquids. Pouring from a graduate cylinder would not be accurate enough and the titration results would be subject to lots of error. A volumetric pipet will not allow you to keep track of the volume needed to complete the titration.
needed to complete the titration. A watch glass and spatula would be inappropriate for this situation because you are dealing with liquids. Pouring from a graduate cylinder would not be accurate enough and the titration results would be subject to lots of error. A volumetric pipet will not allow you to keep track of the volume needed to complete the titration.
In a titration, you should use a buret to administer the 


← Didn't Know|Knew It →
An experiment calls for measuring  of a solution in a graduated cylinder. How should the graduated cylinder be read?
of a solution in a graduated cylinder. How should the graduated cylinder be read?
An experiment calls for measuring 
Tap to reveal answer
In order for an accurate reading of a graduated cylinder, hold the graduated cylinder at eye level then make sure the bottom of the meniscus is at the desired volume. Having the graduated cylinder slightly below eye level will result in readings that are higher than they are supposed to be. Having the graduated cylinder slightly above eye level will result in readings that are lower than they are supposed to be. Never place your head on the same level as the table to read a graduated cylinder. If the graduated cylinder tips over for any reason, you will splash the solution held within on your face.
In order for an accurate reading of a graduated cylinder, hold the graduated cylinder at eye level then make sure the bottom of the meniscus is at the desired volume. Having the graduated cylinder slightly below eye level will result in readings that are higher than they are supposed to be. Having the graduated cylinder slightly above eye level will result in readings that are lower than they are supposed to be. Never place your head on the same level as the table to read a graduated cylinder. If the graduated cylinder tips over for any reason, you will splash the solution held within on your face.
← Didn't Know|Knew It →

3.2 g Zn and 8.1 g HCl are placed in an open beaker. As the reaction proceeds, the hydrogen gas is allowed to escape, causing the reaction to go to completion.
Which reactant (if either) is in excess, and how many grams of it remain?
3.2 g Zn and 8.1 g HCl are placed in an open beaker. As the reaction proceeds, the hydrogen gas is allowed to escape, causing the reaction to go to completion.
Which reactant (if either) is in excess, and how many grams of it remain?
Tap to reveal answer
First calculate the number of mols of each reactant:


The ratio of  to
to  is 1:2. So, given that we have 0.048945 mol
is 1:2. So, given that we have 0.048945 mol  , we only need 0.09789 mol
, we only need 0.09789 mol  . Since we have more
. Since we have more  than is needed,
than is needed,  is in excess. How much is in excess?
is in excess. How much is in excess?

Now convert mols of  to grams:
to grams:

First calculate the number of mols of each reactant:
The ratio of 





Now convert mols of 
← Didn't Know|Knew It →
25 mL of water at room temperature is vaporized and heated to 515 degrees Celsius in an open system.

What will the volume of the resulting gas be? Round your answer to the nearest liter.
25 mL of water at room temperature is vaporized and heated to 515 degrees Celsius in an open system.
What will the volume of the resulting gas be? Round your answer to the nearest liter.
Tap to reveal answer
The system is open, so the pressure can be assumed atmospheric, e.g. 1 atm.
Using the ideal gas law,


We need to convert  to kelvin.
to kelvin.

The density of water at room temperature is approximately 1 g/mL, so we have 25 g of water. Convert to mols:

Plugging everything in:

The system is open, so the pressure can be assumed atmospheric, e.g. 1 atm.
Using the ideal gas law,
We need to convert 
The density of water at room temperature is approximately 1 g/mL, so we have 25 g of water. Convert to mols:
Plugging everything in:
← Didn't Know|Knew It →
A cylindrical metal wire, initially at 100 degrees Celsius, is immersed into a bomb calorimeter containing 100 mL of water at 25 degrees Celsius. After some time, the temperature of the system is homogenous at 35 degrees Celsius.
The wire is 135 cm long with a diameter of 0.1 cm.


What is the heat capacity of the metal? Round your answer to the nearest  .
.
A cylindrical metal wire, initially at 100 degrees Celsius, is immersed into a bomb calorimeter containing 100 mL of water at 25 degrees Celsius. After some time, the temperature of the system is homogenous at 35 degrees Celsius.
The wire is 135 cm long with a diameter of 0.1 cm.
What is the heat capacity of the metal? Round your answer to the nearest 
Tap to reveal answer
First we need to calculate the mass of the wire. We will do this by calculating its volume then multiplying by the density.

Next we need to find out how much energy the water gained from the metal wire.

The energy the water gained is equal to the energy the wire lost. This allows us to calculate the heat capacity for the metal:


First we need to calculate the mass of the wire. We will do this by calculating its volume then multiplying by the density.
Next we need to find out how much energy the water gained from the metal wire.
The energy the water gained is equal to the energy the wire lost. This allows us to calculate the heat capacity for the metal:
← Didn't Know|Knew It →
A chemistry student is trying to determine the identity of an unknown gas. If the gas has a molar mass of  at
at  and
and  , what is the density of this unknown gas under these conditions?
, what is the density of this unknown gas under these conditions?
A chemistry student is trying to determine the identity of an unknown gas. If the gas has a molar mass of 


Tap to reveal answer
We are given the pressure, temperature, and molar mass of the gas in the question stem and are asked to determine the density. To do this, we will need to make use of a manipulated form of the ideal gas equation.

To find density, we will need a way to relate the variables given in the question stem to other parameters, namely mass and volume. Since we already have the volume,  , contained in the ideal gas equation, we just need a way to find the mass,
, contained in the ideal gas equation, we just need a way to find the mass,  . We can do this by noting that the number of moles,
. We can do this by noting that the number of moles,  , can also be written as the mass divided by the molar mass,
, can also be written as the mass divided by the molar mass,  . Substituting this in gives us:
. Substituting this in gives us:

And rearranging, we obtain the density:

But we're not done yet! Remember, we also have to convert the units given for pressure and temperature in the question stem into their proper form. Since we need pressure in terms of atmospheres, and there are  in
in  of gas, we can find that:
of gas, we can find that:

Furthermore, we need to convert degrees Celsius into Kelvins.

Now that we have all the units in their correct form, we're finally ready to plug our values into the equation for density:

We are given the pressure, temperature, and molar mass of the gas in the question stem and are asked to determine the density. To do this, we will need to make use of a manipulated form of the ideal gas equation.
To find density, we will need a way to relate the variables given in the question stem to other parameters, namely mass and volume. Since we already have the volume, 



And rearranging, we obtain the density:
But we're not done yet! Remember, we also have to convert the units given for pressure and temperature in the question stem into their proper form. Since we need pressure in terms of atmospheres, and there are 

Furthermore, we need to convert degrees Celsius into Kelvins.
Now that we have all the units in their correct form, we're finally ready to plug our values into the equation for density:
← Didn't Know|Knew It →
The hospital carries 0.9%  solution of sodium chloride in purified
solution of sodium chloride in purified  . The professor asked his student to prepare 500mL of 0.45% percent
. The professor asked his student to prepare 500mL of 0.45% percent solution sodium chloride in purified
solution sodium chloride in purified  . What volumes of the original solution and purified
. What volumes of the original solution and purified  , respectively, are needed to make the new solution?
, respectively, are needed to make the new solution?
The hospital carries 0.9% 




Tap to reveal answer
This is a typical dilution problem. The first step is to recognize that this is a dilution question. Whenever figuring out how to go from a higher concentrated solution to a lower concentrated solution, it is a dilution problem. The main equation to use here is:



or

or

When plugging in the numbers, make sure to assign the correct volumes and concentrations. Think of one side of the equation describing the 0.9%  solution of sodium chloride and the other side describing the 0.45% solution of sodium chloride.
solution of sodium chloride and the other side describing the 0.45% solution of sodium chloride.


 is the amount of the original solution needed to make the new 0.45% solution. However, we need to add water to the solution to dilute it. To find how much water is needed, subtract:
is the amount of the original solution needed to make the new 0.45% solution. However, we need to add water to the solution to dilute it. To find how much water is needed, subtract:
 of water.
of water.
This is a typical dilution problem. The first step is to recognize that this is a dilution question. Whenever figuring out how to go from a higher concentrated solution to a lower concentrated solution, it is a dilution problem. The main equation to use here is:
or
or
When plugging in the numbers, make sure to assign the correct volumes and concentrations. Think of one side of the equation describing the 0.9% 


← Didn't Know|Knew It →
How many significant figures does the number 42,000,000 have?
How many significant figures does the number 42,000,000 have?
Tap to reveal answer
42,000,000 has two significant figures because of the rule that states that trailing zeroes are never significant unless there is a decimal point. For example, if the number given was "42,000,000." it would be eight significant figures.
42,000,000 has two significant figures because of the rule that states that trailing zeroes are never significant unless there is a decimal point. For example, if the number given was "42,000,000." it would be eight significant figures.
← Didn't Know|Knew It →
While conducting an experiment, you need to measure exactly  of a sulfuric acid solution. Which piece of equipment should you use?
of a sulfuric acid solution. Which piece of equipment should you use?
While conducting an experiment, you need to measure exactly 
Tap to reveal answer
A volumetric pipette is made to dispense a specific amount of liquid very accurately. The Mohr pipette, beaker, and graduate cylinder will not measure a specific amount of liquid as accurately as the volumetric pipette.
A volumetric pipette is made to dispense a specific amount of liquid very accurately. The Mohr pipette, beaker, and graduate cylinder will not measure a specific amount of liquid as accurately as the volumetric pipette.
← Didn't Know|Knew It →
Write the number 42,000,000 in scientific notation using the correct number of significant figures.
Write the number 42,000,000 in scientific notation using the correct number of significant figures.
Tap to reveal answer
The correct answer is  . This is found by moving the decimal place to the left 7 times in order to get one digit to the left of the decimal, and the rest of the digits to the right. The answer just has two significant figures because that is the number of significant figures in the original number.
. This is found by moving the decimal place to the left 7 times in order to get one digit to the left of the decimal, and the rest of the digits to the right. The answer just has two significant figures because that is the number of significant figures in the original number.
The correct answer is 
← Didn't Know|Knew It →
Convert the number  into standard notation, with the correct number of significant figures.
into standard notation, with the correct number of significant figures.
Convert the number 
Tap to reveal answer
To convert into standard notation we must move the decimal two places to the right because of the exponent of "2" above the 10. Then we re-write the number with the decimal in the new space. All of the digits that were in the original scientific notation must be accounted for. Thus, our answer must have six significant figures.
To convert into standard notation we must move the decimal two places to the right because of the exponent of "2" above the 10. Then we re-write the number with the decimal in the new space. All of the digits that were in the original scientific notation must be accounted for. Thus, our answer must have six significant figures.
← Didn't Know|Knew It →
Solve. Account for significant figures.
Calculate using the correct number of significant figures:

Solve. Account for significant figures.
Calculate using the correct number of significant figures:
Tap to reveal answer
The answer is 500 because our rules of significant figures state that when two numbers are multiplied our answer can only have as many significant figures as our number with the fewest significant figures. In this case 5.02 has three significant figures, but 100 only has 1 significant figure. Therefore our answer can only have 1 significant figure.
The answer is 500 because our rules of significant figures state that when two numbers are multiplied our answer can only have as many significant figures as our number with the fewest significant figures. In this case 5.02 has three significant figures, but 100 only has 1 significant figure. Therefore our answer can only have 1 significant figure.
← Didn't Know|Knew It →
Solve and account for significant figures.
Calculate using the correct number of significant figures:

Solve and account for significant figures.
Calculate using the correct number of significant figures:
Tap to reveal answer
The answer is 13.18 because the rules for adding and subtracting significant figures state that the answer can only be precise up to the least precise digit from the numbers that were added/subtracted. In this case 2.12 is only precise to the hundredth place, and therefore our answer can only go to the hundredth place.
The answer is 13.18 because the rules for adding and subtracting significant figures state that the answer can only be precise up to the least precise digit from the numbers that were added/subtracted. In this case 2.12 is only precise to the hundredth place, and therefore our answer can only go to the hundredth place.
← Didn't Know|Knew It →